Hypercoagulability can result from a variety of inherited and, more commonly, acquired conditions. Testing for the underlying cause of thrombosis in a patient is complicated both by the number and variety of clinical conditions that can cause hypercoagulability as well as the many potential assay interferences. Using an algorithmic approach to hypercoagulability testing provides the ability to tailor assay selection to the clinical scenario.
It also reduces the number of unnecessary tests performed, saving cost and time, and preventing potential false results. New oral anticoagulants are powerful tools for managing hypercoagulable patients; however, their use introduces new challenges in terms of test interpretation and therapeutic monitoring. The coagulation laboratory plays an essential role in testing for and treating hypercoagulable states. The input of laboratory professionals is necessary to guide appropriate testing and synthesize interpretation of results. INTRODUCTIONHypercoagulability, also known as thrombophilia, describes a group of hereditary and acquired conditions which confer a propensity to develop thrombi in the veins, arteries, or both. Based on current knowledge, antiphospholipid syndrome is the most prevalent hypercoagulable state, followed by factor V Leiden (FVL) mutation, prothrombin gene G20210A mutations, elevated factor VIII, and hyperhomocysteinemia.
Less common disorders include deficiencies in antithrombin, protein C, or protein S.The prevalence of thrombosis is higher in individuals with a personal and/or family history of thrombosis than in the general population. Acquired and hereditary risk factors for thrombophilia are summarized in ,.
Although patients with hypercoagulable risk factors are at a great risk for developing a thrombotic event, not all patients with hypercoagulable risk factors will develop clinically relevant thrombosis; conversely, not all patients with thrombosis will have an identifiable hypercoagulable state ,. Clinical history and sources of interferenceMany assays used in the laboratory evaluation of hypercoagulability are affected by concurrent clinical conditions and medications. Whenever possible, a thorough clinical history including site of thrombosis, previous bleeding or thrombotic events, other medical disorders (i.e., liver, autoimmune, or cardiovascular disease), pregnancy, medications (including but not limited to anticoagulants), and family history should be elicited.
These are essential for appropriate test selection and interpretation of results; sources of interference for specific assays are described in the following sections. Tests should be performed at least 4-6 weeks after an acute thrombotic event or discontinuation of anticoagulant/thrombolytic therapies including warfarin, heparin, direct thrombin inhibitors (DTIs), direct factor Xa inhibitors, and fibrinolytic agents ,. If abnormal results are found during acute illness or anticoagulant therapy, testing should be repeated in a new specimen when the patient is stable and after anticoagulant therapy is discontinued. Alternatively, thrombophilia testing may be delayed until acute clinical conditions have subsided. The exception is DNA analysis for genetic mutations, which is not generally affected by other medical issues or anticoagulant therapy. Algorithmic approach to laboratory testingNo single laboratory test is yet available that can identify all hypercoagulable defects.
Selection of the most informative tests may differ depending on location and type (venous or arterial) of thrombosis. Outlines a testing algorithm to maximize diagnostic potential in patients with thrombophilia while avoiding unnecessary and potentially expensive tests.
Testing should be performed in a step-wise manner beginning with high-yield screening tests followed by appropriate specific confirmatory tests. These comprehensive panels generate multiple test results that can each be affected by a variety of clinical conditions and drugs. Comprehensive narrative interpretation by coagulation specialists is necessary to synthesize test results, correctly interpret them according to the patient's clinical condition, and provide appropriate guidance to clinicians ,. In some patients with thrombophilia, it may be best to test for all recognized hereditary risk factors, both common and uncommon ,.
Antiphospholipid syndromeAntiphospholipid syndrome (APS) is the most common cause of acquired thrombophilia. Antiphospholipid antibodies (APAs) are acquired autoantibodies directed against phospholipid-protein complexes and are present in 3-5% of the general population. APAs are associated with increased risk of both arterial and venous thrombosis and recurrent pregnancy loss ,. APAs can arise spontaneously (primary) or in association with another condition (secondary). Also known as lupus anticoagulants (LA) because of their prevalence in patients with systemic lupus erythematosus (SLE), APAs are extremely heterogeneous and can be directed against a wide variety of anionic phospholipids, including cardiolipin, beta 2 glycoprotein 1 (B2GP1), and cell-membrane phosphatidylserine ,.Diagnosis of APS requires clinicopathologic correlation because both clinical (either proven vascular thrombosis or pregnancy morbidity) and laboratory criteria must be met.
Laboratory diagnostic criteria include positive testing for 1 of the following on 2 or more occasions, at least 12 weeks apart: (1) lupus anticoagulant; (2) anticardiolipin antibodies (IgG or IgM) in medium or high titer; or (3) B2GP1 antibodies (IgG or IgM) in medium or high titer ,. A repeated positive test after a ≥12-week interval is required for diagnosis because transient low-level increases in APA occur in a variety of clinical conditions, including acute phase response, and may not confer increased risk of thrombosis. 1) Lupus anticoagulant testingBased upon consensus criteria from the International Society for Thrombosis and Haemostasis (ISTH), confirmation of LA requires that the following 4 criteria should be met ,. (1) Prolongation of at least 1 phospholipid-dependent clotting test (e.g., activated partial thromboplastin time aPTT, dilute Russell Viper Venom Test DRVVT screen or hexagonal phospholipid neutralization screen; assays are usually performed with low concentrations of phospholipid to improve sensitivity).
(2) Evidence of inhibitory activity in the patient plasma demonstrated by mixing patient plasma with pooled normal plasma (e.g., immediate and incubated mixing study or DRVVT mixing study). (3) Phospholipid dependence of the inhibitor should be demonstrated by shortening of clotting time after addition of more phospholipid (e.g., DRVVT confirmatory ratio, hexagonal phospholipid neutralization ratio, platelet neutralization). (4) Presence of specific factor inhibitors (especially factor VIII inhibitors) and anticoagulant drugs (heparin or DTI) should be excluded ,. In addition to the ISTH guideline (2009) and updated British Committee for Standards in Haematology (2012) guidelines, the Clinical and Laboratory Standards Institute recently published the first LA guideline ,.
Although all guidelines aim to standardize and harmonize methodologies and improve the quality of LA testing, identifying LA remains a diagnostic challenge.Paradoxically, LAs prolong clot-based assays in vitro while predisposing to thrombosis in vivo. In fact, approximately 30% of LA patients will experience thrombosis. In approximately 15% of patients with deep vein thrombosis (DVT), clotting is attributable to LA ,. Because no single test is available to detect LA, laboratory testing for LA consists of a panel of assays following a diagnostic algorithm. To maximize diagnostic potential, at least 2 assays based on different principles should be performed to evaluate for each of the 4 ISTH criteria. If fewer than 4 diagnostic criteria are met but clinical suspicion for LA exists, the panel is interpreted as indeterminate and should be repeated at a later date ,.Acute thrombotic events or acute phase responses with elevated factor VIII can cause false-negative results.
Thrombin time and anti-Xa assays can help to identify anticoagulant effect or specific inhibitors. Commercially available heparin neutralizers can quench heparin concentrations up to 1.0 U/mL; however, similar reagents are not available for DTIs or specific factor Xa inhibitors. Therefore, LA testing should not be performed on individuals taking these drugs. Individuals on long-term vitamin K antagonist therapy should be tested at least 1-2 weeks after discontinuation of therapy and after the international normalized ratio has normalized to.
2) Anticardiolipin and B2GP1 antibody testingAnticardiolipin antibodies recognize a complex of cardiolipin, a naturally occurring phospholipid, bound to B2GP1 protein. Specific antibodies against cardiolipin and B2GP1 (IgG or IgM) are measured by commercially available solid-phase ELISA. Assays for anticardiolipin antibodies are generally considered sensitive; however, because the antigen target of anticardiolipin antibodies is a B2GP1-cardiolipin complex, B2GP1 antibody assays are considered more specific than anticardiolipin antibody assays ,. It is recommended that both anticardiolipin and B2GP1 APA assays be performed to maximize sensitivity and specificity. Current guidelines include only medium and high levels of antibodies (99th percentile or 40 IgG or IgM phospholipid units) as diagnostic criteria to improve the test specificity.
Individuals with high-titer IgG anticardiolipin antibodies (40 IgG phospholipid units) have been found to have a rate of thrombosis of 6.1% per year, compared with 0.95% per year in individuals with no history of thrombosis, 4.3% in patients with SLE, and 5.5% in patients with a history of thrombosis ,.Positive tests should be repeated after an interval of at least 12 weeks because transient APAs can occur in relation to infection or drugs; these transient APAs are not associated with thrombotic risk ,. Additionally, false-positive results for anticardiolipin antibodies can be associated with a high level of rheumatoid factor and cryoglobulins ,. Activated protein C resistance and FVL mutationProtein C is a vitamin K-dependent serine protease primarily synthesized in an inactive form by the liver. Activation of protein C (APC) requires interaction with the thrombin-thrombomodulin-endothelial protein C receptor complex. Activated protein C regulates thrombin generation by degradation of activated coagulation factors Va and VIIIa in the presence of its cofactor protein S. APC resistance (APC-R) is observed in approximately 20% of patients with a first episode of DVT and 50% of familial thrombosis. More than 90% of APC-R patients have a point mutation in the factor V gene, known as FVL mutation ,.
The FVL mutation (G1691A) results in substitution of glutamine for arginine at position 506 (R506Q); this arginine is 1 of the 3 arginine sites (R306, R506, and R679) cleaved by APC. FVL is present in a heterozygous form in approximately 3-5% of the general Caucasian population and is rare in African, Australian, and South Asian populations ,. The FVL mutation is the most commonly known hereditary risk factor for venous thrombosis (VT); however, risk for arterial thrombosis remains unclear. VT risk is increased 4- to 8-fold in FVL heterozygotes and 80-fold in homozygotes ,.
Risk of pulmonary embolism (PE) may not be as high as risk of DVT. Thrombotic risk is further increased in the presence of a second risk factor: female FVL heterozygotes using oral contraceptives (OCPs) appear to have a 30- to 60-fold increased risk of thrombosis. Other FV mutations occur but are much less common, including FVR2 haplotype (H1299R), FV Liverpool (I359T), FV Cambridge (R306T), and FV Hong Kong (R306G). These affect APC-R and thrombotic risk ,.Acquired APC-R can be caused by the development of autoantibodies against factor V following exposure to bovine thrombin or with untreated hematological malignancies, LAs, pregnancy, OCPs, active thrombosis, elevated factor VIII, and mutations in the factor VIII gene ,.Laboratory assays for APC-R and FVL mutation include functional assays and genotyping.
The functional APC-R assay is based on prolongation of aPTT by degradation of factors Va and VIIIa by exogenously added APC. The ratio of aPTT in patient plasma and normal plasma before and after adding APC is calculated. This ratio in normal individuals is ≥2.0; in FVL heterozygotes, 1.5-2.0; and in homozygotes.
Prothrombin gene G20210A mutationThe prothrombin gene G20210A mutation is a gain of function mutation. The mutation occurs in an intron near the 3' end of the gene and alters 3' end processing and/or enhances translation efficiency, resulting in increased prothrombin (factor II) levels.
However, the exact mechanism of how increased prothrombin gene expression causes hypercoagulability remains unclear. The prothrombin gene G20210A mutation is the second most common hereditary risk factor for VT ,.
Prevalence varies by ethnicity; 2-4% of Europeans carry the mutation, but it is rare in Asians, Native Americans, and Africans ,. This mutation is present in approximately 1-3% of the general population, 5-10% of patients with VT, and up to 20% of patients with VT from thrombophilic families. Heterozygous individuals show a 3-fold increased VT risk. However, VT risk will be drastically increased when the patients carry additional inherited or acquired risk factors ,.Genetic testing to detect the G20210A mutation can be performed by PCR-based methods.
Newer assays based on various PCR methods coupled with fluorescence polarization methods or the Invader assay can be performed on automated platforms. DNA microarray technology can detect multiple genetic markers simultaneously with a relatively low cost as a single test compared with conventional DNA assays ,. However, these tests require expensive equipment and skilled personnel and may require reflex confirmatory assays (sequencing) if there is an ambiguous or atypical result. Protein C deficiencyProtein C deficiency occurs in 0.14-0.50% of the general population and 1-3% of patients with VTE ,.
It is inherited in an autosomal dominant fashion; heterozygotes show functional protein C levels of 40-65% of normal and have a 7-fold increase in risk for VT. The first thrombotic event usually presents at 10-50 years of age. Protein C deficiency also carries increased risk for warfarin-induced skin necrosis. Homozygotes are very rare and can present with neonatal purpura fulminans or disseminated intravascular coagulation (DIC).Protein C assays measure activity (functional) or antigen quantity (immunological). A functional protein C assay, either clot-based or chromogenic, is usually performed first; if the result is low, an antigenic protein C assay is performed to determine whether the protein defect is quantitative (type I) or qualitative (type II). Type I protein C deficiency is characterized by reduced functional activity and antigen levels and is much more common (75-80%) than type II deficiency (20-25%), which is further subdivided into types IIa (24.5%) and IIb (0.5-1.0%). Both IIa and IIb result in reduced activity with normal antigen levels.
If only a quantitative antigenic assay is used, type II deficiency cannot be detected ,. Clot-based functional protein C assays can detect both types I and II deficiencies but can give falsely increased results with anticoagulant therapy, LAs, and FVL mutation, and falsely decreased results with elevated factor VIII levels (particularly 250%) or low protein S.
The chromogenic assay is less affected by interfering substances and is more reproducible; however, it only assesses alterations in the activation and active sites of the protein (type IIa defects) and cannot detect defects in other sites (protein S, surface, or substrate binding sites) and therefore can overlook the rarer type IIb deficiencies ,. Functional assays, either chromogenic or clot-based, are recommended as initial screens with antigenic assays performed if results are abnormally low.Acquired deficiency is more common than hereditary forms and must be excluded before making a diagnosis of hereditary protein C deficiency.
Because protein C is synthesized in hepatocytes and is vitamin K-dependent, both liver dysfunction and vitamin K deficiency (including warfarin therapy) decrease protein C levels. Protein C has a short half-life (6-8 hours), and levels decrease more rapidly than other coagulation proteins (including protein S and antithrombin) in liver disease and warfarin therapy/vitamin K deficiency. Conversely, protein C levels rapidly normalize after discontinuation of warfarin or correction of vitamin K deficiency; however, levels should not be measured for at least 10 days after warfarin discontinuation. Protein C levels are lowered in recent or current thrombosis, DIC, L-asparaginase therapy, and nephrotic syndrome and during the intra- or immediately postoperative period; neonates also have relatively low protein C levels (17-53%). OCP use and pregnancy can increase protein C levels ,.
Abnormal protein C assays should be repeated after any such conditions have resolved to confirm results. Protein S deficiencyProtein S is a vitamin K-dependent glycoprotein that acts as a cofactor to protein C, accelerating proteolysis of factors Va and VIIIa by approximately 2-fold. Approximately 60% of protein S in the plasma is bound noncovalently to C4bBP in plasma with high affinity; the remaining free (unbound) protein S is the predominant active form.
Recent studies have shown that protein S also exerts its own anticoagulant activity by direct binding of factors V, VIII, and X, and appears to act as a cofactor for the tissue factor pathway inhibitor, which results in inhibiting tissue factor-mediated factor X activation ,. Hereditary protein S deficiency is transmitted in an autosomal dominant fashion and occurs in 0.2-0.5% in the general population and in 1-3% of patients with first VT ,.
Functional protein S levels range 20-64% in heterozygous patients. Homozygous patients typically present as newborns with purpura fulminans and DIC.There are 3 types of protein S deficiency. Types I and III are quantitative deficiencies with both low free protein S antigen and low protein S activity, and account for 95% of cases. Type I deficiency shows low total (free+C4bBP-bound) protein S antigen level, whereas type III shows normal total protein S levels. Type III deficiency may be related to excess binding of protein S to C4bBP.
Type II deficiency is a qualitative defect with low protein S activity and normal antigenic (free and total) protein S levels ,.Functional (clot-based) protein S assays measure activity, and immunologic methods (monoclonal antibody-based enzyme immunoassay and immunoturbidimetric assay) measure antigenic levels of free or total protein S. Clot-based functional assays are sensitive for all types of deficiency and are used by some laboratories as an initial screening test, similar to the algorithm for protein C. However, functional protein S assays are not specific; potential sources of interference include APC-R (usually from FVL mutation) and LAs. Despite the fact that using an antigenic test as an initial assay may miss type II protein S deficiency, some guidelines recommend antigenic tests as a screen with functional tests performed only on abnormal results.As with protein C deficiency, acquired causes of protein S deficiency are more common and should be excluded before making a diagnosis of hereditary deficiency.
Protein S is decreased in conditions that decrease protein C (see the previous section). Protein S is also decreased during the acute phase response because C4bBP is an acute phase reactant; increased C4bBP lowers both protein S activity and free antigen. Protein S is decreased with elevated factor VIII (250%), and infectious and autoimmune conditions such as HIV infection and inflammatory bowel disease ,. Levels are usually lower in women, especially during hormone replacement therapy, OCP use, and the second or third trimester of pregnancy. Abnormal results should be confirmed after any such conditions are resolved. Antithrombin deficiencyAntithrombin (half-life: 2-3 days) is a glycoprotein of the serine protease inhibitor (serpin) family that primarily inactivates activated thrombin (factor IIa) and factor Xa, and to a lesser extent, factors IXa, XIa, and XIIa. Antithrombin acts as a 'suicide inhibitor' by forming 1:1 covalent complexes between antithrombin and serine proteases; inhibitor activity is greatly accelerated by interaction with heparin.
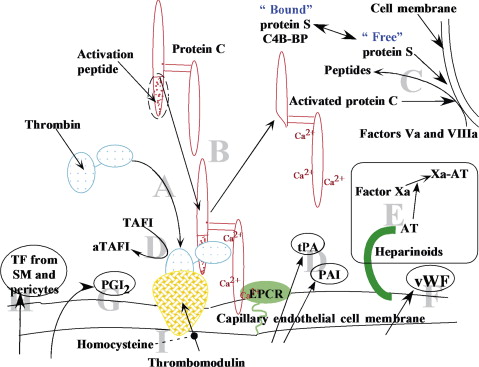
Although synthesized in the liver parenchyma, antithrombin is not vitamin K-dependent ,.Antithrombin deficiency is inherited in an autosomal dominant fashion and associated almost exclusively with VT. Prevalence rates are approximately 0.05-0.1% in the general population. Estimated annual incidence of a first episode of VTE in heterozygotes is 1.0-2.9% annually in retrospective studies ,. Risk of thrombosis appears to be higher in antithrombin deficiency than protein C or protein S deficiency, APC-R, or prothrombin gene G20210A mutation and thus has the highest VTE risk among known hereditary thrombophilias.
The homozygous state is almost universally fatal in utero. Functional antithrombin levels in heterozygous individual ranges 35-70% ,. Besides presenting as DVT or PE, VTE from antithrombin deficiency can occur in unusual sites, such as cerebral sinuses and the mesenteric, portal, and renal veins. The first event occurs at a young age (. HyperhomocysteinemiaHomocysteine is an intermediate amino acid produced by demethylation of methionine via methylenetetrahydrofolate reductase (MTHFR) in the folate cycle.
Homocysteine metabolism requires vitamin B6, vitamin B12, and folate. Hyperhomocysteinemia is associated with increased risk of VTE, coronary heart disease, acute myocardial infarction, peripheral artery disease, stroke, aneurysm, migraine, hypertension, male infertility, risk for offspring with neural tube defect, and recurrent pregnancy loss. Acquired hyperhomocysteinemia can be caused by vitamin B6, vitamin B12, or folate deficiency; renal failure; hypothyroidism; rheumatoid arthritis; and certain drugs such as methotrexate, niacin, anticonvulsant, theophylline, L-dopa, thiazide, cyclosporine A, or phenytoin ,.Hereditary hyperhomocysteinemia is caused by polymorphisms in an enzyme necessary in homocysteine conversion pathways. Homozygous alterations of the MTHFR gene are present in 10-13% of the population, whereas heterozygous alterations are found in 30-40%. Two commonly recognized polymorphic variants in the MTHFR gene are (1) the 'thermolabile' c.665C-T (p.Ala222Val), historically referred to as c.677C-T, and (2) c.1286A-C (p.Glu429Ala), also known as c.1298A-C. Meta-analyses of 2 common polymorphisms have found a weak association with risk of thrombosis ,. Homozygosity for MTHFR c.665C-T is associated with an approximately 25% increase in plasma homocysteine.
Patients with elevated homocysteine and the MTHFR c.665C-T and/or c.1286A-C mutations, however, may be at mildly increased risk for both VTE (odds ratio: 1.27) and recurrent pregnancy loss (pooled risk: 2.7). Lowering homocysteine levels using vitamin B6, vitamin B12, or folate has not been proven to reduce thrombotic risk.
Given the modest increase in thrombophilic risk and lack of evidence of therapeutic benefit, screening homocysteine levels in healthy individuals and testing for MTHFR polymorphisms are not currently suggested ,. Elevated factor VIIISeveral studies have demonstrated an association between elevated factor VIII and increased risk of thrombophilia, partly due to factor VIII-mediated enhancement of thrombin generation ,. Although no genetic variations in the factor VIII gene have been identified, levels appear to be higher in African-Americans and lower in individuals with blood group O. Factor VIII can be elevated during acute phase reactions, elevated estrogen, pregnancy, or after aerobic exercise. The prevalence of elevated factor VIII among patients with VT is 20-25% ,. Whether factor VIII elevation directly contributes to increased thrombophilic risk remains unclear; however, studies show that persistent factor VIII level 150% (or 90th percentile) in the absence of acute phase reactions, elevated estrogen levels, or recent exercise is an independent risk factor for thrombophilia ,.Factor VIII activity can be measured by aPTT-based clotting assay or chromogenic assay, and antigen quantitation can be accomplished using ELISA. Factor VIII measurement should be postponed until at least 6 months after an acute thrombotic event and 6 weeks after giving birth and should be repeated after 3-6 months to confirm persistent elevation ,.
Fibrinogen defectsDysfibrinogenemia describes a heterogeneous group of disorders resulting in structurally and functionally altered fibrinogen. It can cause bleeding, venous or arterial thrombosis, or both. The prevalence of dysfibrinogenemia in patients with VT is approximately 0.8% ,. Although the exact mechanism of thrombosis is unknown, it may be related to increased fibrin formation and/or impaired fibrinolysis. Patients with dysfibrinogenemia may have prolonged prothrombin time (PT), thrombin time, and reptilase time; decreased functional fibrinogen; and normal to elevated immunologic fibrinogen. The ratio of functional fibrinogen activity to immunologic fibrinogen antigen will be decreased ,.The most commonly used functional assay is the Clauss method. Acquired fibrinogen deficiency can be caused by liver disease, consumptive states such as placental abruption or DIC, or fibrinolytic therapy.
Because fibrinogen is an acute phase reactant, testing should be delayed at least 6 months after acute thrombosis. MONITORING DIRECT ORAL ANTICOAGULANTSWarfarin has been the only orally administered anticoagulant and thus the mainstay of outpatient management of patients with hypercoagulable states. Although effective, warfarin has multiple interactions with medications and food and variable pharmacogenetics that necessitate routine monitoring.
Patients appear to spend only a fraction of time in the therapeutic range, leaving them at risk both for thrombosis and bleeding. Recently, new direct oral anticoagulants (DOACs) have been introduced, including the direct thrombin inhibitor dabigatran and direct factor Xa inhibitors rivaroxaban and apixaban, which have more predictable pharmacodynamics and pharmacokinetics than warfarin.
They are also characterized by a rapid onset of anticoagulant activity and fixed dosing with peak blood concentrations 2-4 hours after ingestion. Notably, none currently has an antidote or specific reversal agent. These drugs were initially shown to be noninferior to warfarin for prevention of stroke and systemic embolism in patients with nonvalvular atrial fibrillation ,. Utorrent free download.
Since then, all 3 have also been approved in the United States for VTE prophylaxis in patients undergoing hip and knee replacement, and dabigatran and rivaroxaban are approved for treatment and reduction of risk of recurrence of DVT and PE.This section briefly describes recommendations for monitoring DOACs. For a more in-depth discussion, readers are directed to the published ISTH Guidelines and recent reviews by Drs.
Eby and Mani, Kasper, and Lindhoff-Last ,. These reviews also describe the influence of DOACs on other coagulation assays, including LA testing, which is not covered here.Although most patients being treated with DOAC do not require monitoring, there are certain clinical scenarios in which measurement of drug levels is necessary.
These include episodes of bleeding, before surgery or invasive procedure and perioperative management, concomitant use of drugs with known DOAC interactions, body weight extremes, renal impairment, suspected overdose or noncompliance, and DOAC treatment failure (thrombosis during therapy). For emergent situations, especially if drug history is not known, or at peak drug levels, a qualitative or semiquantitative assay will often suffice. However when dose adjustment is necessary (e.g., during treatment failure, renal impairment), a quantitative assay is required ,.Although dabigatran and factor Xa inhibitors influence routine PT and aPTT, the effects are variable, with dabigatran exerting more influence on aPTT and factor Xa inhibitors prolonging PT. Moreover, the degree of prolongation is highly dependent on the reagent used for the assay. These widely available tests can be used to detect peak or supratherapeutic drug levels, but should not be used for quantitation. They may also appear normal during low or trough drug levels.
Each laboratory should be aware of how its specific PT and aPTT reagents behave in the presence of these drugs. This can be achieved using commercially available calibrators ,.Because of its mechanism of action, the thrombin time (TT) is exquisitely sensitive for dabigatran, and a normal TT essentially excludes the presence of the drug. The effect on TT is linear, but for monitoring near the therapeutic range, a diluted assay is necessary. The ecarin clotting time or chromogenic anti-IIa assays can also be used to quantitate DTI effect; however, these tests are not widely available ,.For anti-Xa inhibitors, the PT can be used as a qualitative assay; in addition, the STA Neoplastin Plus reagent (Diagnostica Stago, Asnieres, France) can reportedly be used for quantitative measurement of rivaroxaban. However, the PT is generally less sensitive for apixaban than rivaroxaban. Anti-Xa assays, chromogenic or clot based, are widely available because they are used in heparin and heparin-like drug monitoring.
These assays can be used to quantitatively and sensitively measure factor Xa inhibitor effects. Although interlaboratory variability has been reported in the past, commercially available calibrators are now available to aid in standardization ,. Summarizes the recommended assays for different types of DOAC monitoring.
An Algorithmic Approach To Hemostasis Testing Freeze
The global incidence of venous thromboembolism is high so laboratory testing for hereditary thrombophilia and selected acquired thrombophilia is common. Given the costs associated with testing and multiple pre‐analytic and analytic variables affecting the assays, careful patient selection and timing of testing and diligent application to patient management are critical to providing high‐value clinical care. Collaboration between the ordering providers and performing laboratories has the potential to achieve these goals. Herein, utility of thrombophilia testing, variables that affect the assays, and impact on patient management are reviewed. Where available, information on cost‐effectiveness is discussed.
1 IntroductionIt is estimated that, in the United States (US), the incidence of deep vein thrombosis and pulmonary embolism (venous thromboembolism, VTE) is approximately 1.92 per 1000 person‐years and accounts for approximately 1% of hospital admissions. The global disease burden is similar, estimates range from 0.75 to 2.69 per 1000 individuals in the population, but increases with age. Venous thromboembolism is a multifactorial disease; this means that patients with hereditary thrombophilia (e.g, factor V Leiden) may go through their lifetime without experiencing VTE, but when exposed to an acquired risk factor (e.g, orthopedic surgery of knee) may develop a VTE. Thus, special coagulation assays, to determine the presence or absence of an underlying hereditary thrombophilia, are commonly ordered. The term thrombophilia connotes the presence of a risk factor for VTE which may be hereditary or acquired. The presence of a hereditary thrombophilia does not necessarily imply that the patient will definitely develop VTE, as each thrombophilia is associated with a different degree of risk for developing a first lifetime and recurrent VTE (Table ). Multiple hereditary abnormalities in coagulation proteins, for example, plasminogen activator inhibitor‐1, have been described but in isolation are felt to be weak risk factors for VTE and laboratory testing is not recommended.
Similarly, the multiple acquired thrombophilias put patients at variable risk during periods of exposure; however, most such risk factors are considered to be reversible or temporary, except for persistent antiphospholipid antibodies (Table ). Although the risk of a first lifetime VTE with antiphospholipid antibodies (lupus anticoagulant and/or anticardiolipin and/or beta 2 glycoprotein I antibodies) is not well defined, they are associated with an increased relative risk of recurrent VTE, odds ratio 1.41 (CI 0.99‐2.00). Several recent publications have reviewed details of laboratory aspects of thrombophilia testing -; herein, clinical utility, selected laboratory aspects, and an approach to optimal utilization of thrombophilia testing are discussed. 2 Clinical Utility, Cost‐Effectiveness, and Impact on Patient Management of Thrombophilia TestingDebates on the “pros and cons” of thrombophilia testing are ongoing,; however, there is likely not a single correct approach. The utility of testing will vary with the population under consideration, for example, asymptomatic individuals, asymptomatic individuals at risk for disease (VTE), and symptomatic patients with VTE. A second important consideration is whether to perform a panel or profile of tests or to focus on individual assays. In addition to its diagnostic role, determining an underlying thrombophilia has played a prognostic role (e.g, predicting risk of recurrent VTE) which may or may not influence patient management.
In the current environment of constrained healthcare resources, a bigger emphasis needs to be placed on whether and how testing will impact patient management. Well‐designed studies have demonstrated that not all thrombophilias predict a higher risk of recurrent VTE, thus raising questions regarding utility of testing. Pretest counseling for hereditary thrombophilia is important and should encompass an understanding of the overall risk of VTE, risk associated with exposure to acquired thrombophilia, impact on management, and the potential impact on being labeled with a hereditary disorder. In addition, there are unique aspects to counseling women given their exposure to estrogens and pregnancy. 3.1 Activated protein C resistance/factor V Leiden and prothrombin G20210AActivated protein C resistance due to the factor V Leiden (FVL) mutation is the most common hereditary thrombophilia among the white population. The FVL carrier frequency ranges from 2% in Southern Europe to 15% in southern Sweden.
About 3‐7% of asymptomatic U.S. White populations of northern European or Scandinavian ancestry are heterozygous carriers, but a lower carrier frequency among nonwhites, 1.2% in African Americans, 2.2% in Hispanics, 1.2% in Native Americans, and 0.45% in Asian Americans. The ethnic distribution of the prothrombin G20210A (PT G20210A) is similar to that of FVL. The annual incidence of VTE in the general population and heterozygous carriers of APCR/FVL is approximately 0.1% per year and 0.45%, respectively, and the estimates of developing VTE by age 65 years vary from 2.4% to 6%. This high prevalence not only makes it the most commonly tested thrombophilia but one that has been well studied, especially the interactions with acquired thrombophilia.Although the relative risk of oral contraceptive pill (OCP)‐associated VTE in APCR/FVL heterozygotes is estimated to be increased by fivefold to 30‐fold, in absolute terms, the incidence of VTE increases from a baseline risk of 1 per 10 000 women‐years in those not on OCP to 30 per 10 000 women‐years among those on OCP or approximately 0.3% per year. During pregnancy, the incidence of VTE among noncarriers is 0.73 per 100 pregnancy‐years and increases to an incidence of 1.97 per 100 pregnancy‐years for heterozygous carriers. Given the low absolute risks of VTE, routine APCR/FVL screening pre‐OCP was not found to be cost‐effective and prepregnancy screening is discouraged.
There are no similar detailed data for the PT G20210A mutations; however, heterozygous carriers on OCP have an approximate sixfold increased risk for VTE and 150‐fold increased risk of cerebral vein thrombosis. As VTE incidence increases with age, there is a higher population prevalence of VTE among postmenopausal women, and when exposed to (estrogen) hormone replacement therapy (HRT), APCR/FVL heterozygotes have an odds ratio of VTE up to 15‐fold higher when compared to noncarriers, or an annual absolute incidence of 1% in this population of patients. Based on the high absolute risk of VTE, compared to no screening, APCR/FVL screening prior to HRT use was found to be cost‐effective if HRT was only dispensed to noncarriers. Surgery (orthopedic and nonorthopedic) is a significant risk factor for VTE (Table ); however, routine screening for APCR/FVL, to tailor VTE prophylaxis, was not found to be cost‐effective.
3.2 Clinical utility of testing for AT, PC, and PS deficiencyThe prevalence of hereditary AT, PC, or PS deficiencies in the white and nonwhite population is. 3.3 Clinical utility of testing for MTHFR variantsMild hyperhomocysteinemia is a weak risk factor for VTE. Although hyperhomocysteinemia may be due to MTHFR mutations, testing for the MTHFR C677T and A1298C variants, especially in the presence of a normal plasma homocysteine level, provides no clinical utility and is not indicated. The American College of Medical Genetics (ACMG) practice guidelines recommend against MTHFR polymorphism genotyping for a thrombophilia or recurrent pregnancy loss. In addition, genotyping should not be performed on at‐risk family members.
4 Thrombophilia Testing in Venous ThromboembolismThrombophilia testing in patients with VTE is reasonable; however, circumstances of VTE, timing of testing, and impact on patient management should guide this decision. Thrombophilia testing not indicated for patient managementPopulation screeningVTE occurring in association with temporary risk factorPatients currently on therapeutic anticoagulationPatients with arterial thrombosis aLupus anticoagulant testing is indicated; VTE, venous thromboembolism.Patients on whom long‐term therapeutic anticoagulation is plannedTo tailor VTE prophylaxis in high‐risk situations bFor example, duration of prophylactic anticoagulation after hip or knee replacement surgery.Thrombophilia testing is reasonableYoung individuals with VTE, for example. 5.2 Available assaysTesting for APC resistance (APCR) can be performed using plasma‐based functional or by direct DNA‐based assays for FVL (the most common inherited cause of APCR). The functional assay is the preferred initial test, which if abnormal can be followed up with the DNA‐based assay to confirm genotype. There can be situations where there are discrepancies between the two assays (e.g, one factor V allele not expressed) or in FVL carriers who have APCR, who receive a hematopoietic stem cell transplantation from a noncarrier; the patient may be misdiagnosed as being unaffected. In contrast, a FVL carrier may receive a liver transplant from a noncarrier and may be considered to be affected if FVL testing was the sole method of testing.
A commonly used assay is one in which the patient plasma is mixed with FV‐deficient plasma, thus correcting for potential deficiencies of other coagulation factors (e.g, liver disease, warfarin anticoagulation effect, vitamin K deficiency), potentially diluting the effect of lupus anticoagulants; in addition, some assays are validated to neutralize up to 1 U/mL of heparin in the plasma sample (manufacturer's package insert). When optimized, the assay is essentially 100% sensitive and specific for FVL, and commonly, results for heterozygotes and homozygotes are nonoverlapping. 5.3 Variables affecting APCR assaysThe presence of oral direct thrombin and Xa inhibitors may result in a false‐normal APCR, and collection of the specimen in EDTA may result in a falsely reduced APCR ratio, and this assay may not detect acquired APCR (Table ). Additional available options for APCR testing have recently been reviewed; each laboratory must establish or validate reference ranges given the variable instrumentation. A variety of methods for direct DNA‐based testing for FVL are available for diagnosis; commonly used molecular methods involve PCR amplification of the region surrounding the mutation, followed by restriction enzyme digestion (RFLP), allele‐specific PCR amplification, or allele‐specific hybridization.
Semi‐automated assay methods include fluorescence detection of PCR products with allele‐specific hybridization probes, and non‐PCR signal amplification methods based on either enzymatic hybridization mismatch recognition using fluorescent allele‐specific probes or linked fluorescent allele‐specific pyrophosphorolysis‐kinase reactions. Each laboratory must insure that their method can distinguish factor V Leiden from an uncommon polymorphism A1696G. 6 Prothrombin G20210GA MutationThe PTG20210A affects the 3′ terminal nucleotide of the 3′ untranslated region of F2 and is associated with increased protein synthesis,; however, despite higher median plasma prothrombin levels, which is the basis of the phenotype, in carriers compared to noncarriers of the mutation, the overlap of the range of prothrombin levels precludes diagnosis with plasma‐based assays; thus, molecular testing is the only option for diagnosis of this thrombophilia disorder. 7.1 Diagnosis and classificationThe diagnosis relies on plasma‐based functional and antigen assays.
The deficiencies are classified into type I or quantitative (decreased functional and antigen assays) and type II or qualitative (decreased functional and normal antigen assays) defects. In type III PS deficiency, the PS activity and free PS antigen are reduced and total PS antigen is normal. Type II AT deficiency is subclassified as follows: IIa: reactive site (RS) mutations, IIb: heparin‐binding site (HBS) mutations, and IIc: pleiotropic effect (PE) mutations. 7.2 Available assays for protein C, protein S, and ATProtein C activity assays may be clot‐based or chromogenic.
The functional assays (PTT‐based and RVV‐based) have different performance characteristics., Although using a clot‐based assay will likely detect most variants of congenital protein C deficiency, multiple pre‐analytic and analytic interferences may result in artifactual results (Table ). Use of chromogenic assays (which has less potential for interference) may miss small percentage of congenital protein C deficiency., For protein S testing, functional assays (based on PTT, PT, Xa, and RVV) are available but are fraught with variables that affect results. Antigenic assays (for free and total PS antigen) are more reliable and measure free protein S (approximately 40% of total plasma protein S) and total protein S antigen (which includes the 40% free protein S and 60% that is bound to C4B‐binding protein). Functional assays for antithrombin are generally chromogenic assays and are based on inhibition of factor IIa (thrombin) or factor Xa.
Page/Link:Page URL:HTML link:The Free Library. Retrieved Aug 27 2019 fromPlatelets are small (2-microm-diameter), nonnucleated blood cellsproduced in the bone marrow from megakaryocytes. Platelets are activatedrapidly after blood vessel injury or blood exposure to the artificialsurfaces of implanted devices, and they are a crucial component of theprimary hemostatic response. In their inactivated state, platelets areroughly discoid in shape and contain cytoplasmic organelles,cytoskeletal elements, invaginating open-canalicular membrane systems,and platelet-specific granules, called alpha and dense granules.Platelets have numerous intrinsic glycoproteins embedded in the outersurface of their plasma membrane that are receptors for ligands rangingfrom fibrinogen, collagen, thrombin, and thrombospondin to vonWillebrand factor (VWF) and fibronectin. (1-3) Platelets promotehemostasis by 4 interconnected mechanisms: (1) adhering to sites ofvascular injury or artificial surfaces, (2) releasing compounds fromtheir granules, (3) aggregating together to form a hemostatic plateletplug, and (4) providing a procoagulant surface for activated coagulation protein complexes on their phospholipid membranes (Figure 1).FIGURE 1 OMITTEDPlatelet adhesion to the subendothelium is the initial step inplatelet activation. The subendothelium is composed of extracellularmatrix proteins, such as collagen, fibronectin, VWF, thrombospondin, andlaminin, (4) many of which are ligands for receptors on the plateletsurface.
These adhesive proteins are exposed when the endothelial layeris disrupted. Because of the large number of extracellular matrixproteins and a high density of platelet surface receptors, plateletadhesion to areas of vascular injury is extremely rapid. VWF, a large,multimetric protein secreted into the extracellular matrix fromendothelial cells, facilitates platelet adhesion by binding to plateletsurface glycoprotein Ib/IX/V, especially at high shear rates. (5-7)Platelets can also adhere to vascular wall-associated fibrin orfibrinogen through interaction with platelet surface glycoproteinIIb/IIIa.
After adhering to the subendothelium, platelets undergo acytoskeletal activation that leads to a shape change with development ofpseudopods. Intracellular signaling processes lead to increasedcytoplasmic calcium and then initiate a secretory release reaction,whereby products from the alpha granules (platelet factor 4,beta-thromboglobulin, thrombospondin, platelet-derived growth factor,fibrinogen, VWF) and dense granules (adenosine diphosphate ADP,serotonin) are released into the surrounding milieu.
(10) The granule membranes contain many integral glycoproteins on their inner leaflet,such as P-selectin (CD62p) in the alpha granule and gp53 (CD63) in thelysosome, which become expressed on the outer platelet membrane afterthe release reaction. (11) The release of ADP from the dense granules,together with calcium mobilization, leads to a conformational change ofthe fibrinogen receptor, the glycoprotein IIb/IIIa receptor complex(integrin alpha.sub.IIbbeta.sub.3). (12) This conformationalchange of the fibrinogen receptor initiates the process of aggregation,whereby a glycoprotein IIb/IIIa receptor on one platelet is bound in ahomotypic fashion to the same receptor on adjacent platelets via acentral fibrinogen molecular bridge. Beside ADP, other agonists, such asepinephrine, thrombin, collagen, and platelet-activating factor, caninitiate platelet aggregation by interaction with membrane receptors.This platelet release reaction and aggregation lead to the recruitmentof many other platelets to the vessel wall with the formation of ahemostatic platelet plug.Activated platelets also play a vital procoagulant role that servesas a link between platelet function and coagulation activation. Plateletmembrane phospholipids undergo a rearrangement during activation with atransfer of phosphatidyl serine from the inner table to the outer tableof the platelet membrane, providing a binding site forphospholipid-dependent coagulation complexes that activate both factor Xand prothrombin. (13)LABORATORY TESTS USED IN THE EVALUATION OF PLATELET FUNCTIONClinical HistoryA careful clinical and family bleeding history should be takenbefore beginning a laboratory evaluation of platelet function.
Thehistory should include an assessment of the duration, pattern, andseverity of bleeding problems, including whether the bleeding isspontaneous or associated with trauma or surgery. A lifelong bleedingdiathesis may suggest a congenital platelet dysfunction, but an onset inadulthood does not necessarily exclude a congenital problem. Inobtaining a history of bleeding pattern, it is necessary to determinewhether a true hemorrhagic disorder exists.
In this regard, it is oftenhelpful to assess if the bleeding is out of proportion to the degree oftrauma, or whether blood transfusions were required for relatively minorsurgical procedures, such as tooth extractions. Platelet-mediated bleeding disorders usually result in amucocutaneous bleeding pattern, with ecchymosis, petechiae, purpura,epistaxis, and gingival bleeding commonly observed. (4) This pattern isin contrast to that observed with coagulation protein disorders, inwhich deep tissue bleeding and hemarthroses are more common. VonWillebrand disease, an abnormality of VWF, has bleeding symptoms verysimilar to platelet dysfunction, and evaluation for von Willebranddisease should be included in the initial evaluation of a possibleplatelet disorder. (14) Bleeding diatheses due to vascular malformationsmay give a bleeding pattern similar to platelet disorders, but is oftenmore focal than diffuse.
Acquired purpuras, such as those seen withdisseminated intravascular coagulation, vasculitis, or infections, canusually be distinguished from platelet dysfunction, because plateletdisorders usually cause bleeding from mucous membranes ('wet'purpura), whereas vascular purpura is usually confined to the skin('dry' purpura). (15)Many drugs and foods can affect platelet function (Table 1), so acomplete drug history should be obtained.
(16) It is important toremember that aspirin, an irreversible inhibitor of platelet function,is an ingredient of many over-the-counter and prescription medications,such as cold or flu remedies. Platelet dysfunction is associated withmany systemic disorders, such as renal disease, hepatic failure,connective tissue disorders, myeloproliferative disorders,myelodysplastic disorders, malignancy, and cardiovascular disease.Additionally, some clinical features, such as albinism, deafness,nephritis, and susceptibility to infections, may help in thedifferential diagnosis of the inherited platelet disorders.
(17)Platelet Count and Peripheral Blood SmearThe accepted normal range of the platelet count is generallybetween 150 to 400 X 10.sup.3/microL of blood, although values muchlower than this can be quite adequate for hemostasis. Many clinicianswill refrain from platelet repletion in a stable patient until countsdrop below 10 to 20 X 10.sup.3/microL. (18) Initial evaluation ofthe platelet count must take into consideration anypseudothrombocytopenia. Pseudothrombocytopenia is often due tocold-reacting platelet agglutinins or platelet binding to neutrophils (platelet satellitism).
The agglutinins are often seen in patients withhigh immunoglobulin levels or infections and usually only bind plateletswhen calcium is chelated, such as in an EDTA blood collection tube. (19)A pseudothrombocytopenia associated with the glycoprotein IIb/IIIaantagonist drug abciximab has also been reported. (20)Pseudothrombocytopenia can be diagnosed by examining a peripheral smear,where large aggregates of platelets are observed, often around thefeathered edge. A more accurate platelet count can be established bycollecting the blood sample in either citrate or heparin anticoagulants.Giant platelets observed with macrothrombocytopenia syndromes can givefalse low platelet counts, because the large platelets may be counted asleukocytes by automated cell counters.
(21)The mean platelet volume (MPV) is an indication of platelet size.Normal MPV ranges are approximately 7 to 11 fL. The MPV can be anindication of platelet turnover, because platelets newly released fromthe bone marrow are larger and tend to decrease in size with age in thecirculation.
(22) In patients with rapid turnover, the platelets will,in general, be larger because of the larger size of newly producedplatelets. True congenital macrothrombocytopenias usually have uniformlylarge platelets; often the platelets are at least twice the normal sizeand may be as large as erythrocytes. Newer techniques based on messengerRNA detection in platelets (reticulated platelets) may also be helpfulto indicate the rate of thrombopoiesis. (23,24)Platelet disorders can be associated with varying plateletappearances.
In von Willebrand disease, Glanzmann thrombasthenia, andmyeloproliferative disorders, the platelets have typical morphologicfeatures, whereas giant platelets are seen in Bernard-Soulier diseaseand other macrothrombocytopenia syndromes. (17,25) In patients withWiskott-Aldrich syndrome, the platelets may be small. (26) Platelets inthe gray platelet syndrome, an alpha granule deficit, are characteristicfor being pale, gray, and hypogranular on a Wright-stained blood smear.(27) Some platelet storage pool disorders (SPDs) may havemorphologically normal platelet counts by light microscopy, but may havedecreased alpha and/or dense granules by electron microscopy.
(28)Platelet Function Screening Tests or Bleeding TimeIn the initial evaluation of platelets, it is desirable to performa screening test to evaluate platelet function. For nearly a century,the bleeding time was the only platelet function screening testavailable. (29) The bleeding time is a test that is fraught withvariability and involves the creation of a standardized cut in the skinand measurement of the time it takes for bleeding to stop.
The initialDuke bleeding time used a small incision in the earlobe, and the similarRatnoff method uses an incision in the ball of the finger. (29,30) Themost commonly used bleeding time is the Ivy bleeding time, where astandardized incision is made on the volar surface of the forearm with aspring-loaded device, using venostatic pressure applied on the upper armby a sphygmomanometer. (31) The bleeding time result depends not only onplatelet number and function, but also on fibrinogen concentration,adequate vascular function, orientation and size of the incision, siteof the incision, skin quality, skin temperature, operator technique, andpatient cooperation.Although procedural variability affects the bleeding times,bleeding time has been included traditionally as a screening test forsuspected bleeding disorders. (32) The bleeding time has little use as apresurgical screen for hemostatic competence in individuals without ahistory of bleeding and is not useful in discerning platelet dysfunctionin thrombocytopenic patients. (33) Many laboratories have stoppedperforming the bleeding time test completely because of its variability,poor reproducibility, and lack of correlation with intraoperativebleeding. Newer automated whole blood platelet function screeningassays, such as the Platelet Function Analyzer-100 (PFA-100, DadeBehring, Marburg, Germany), are gaining popularity as initial screensfor platelet function even though they do not measure the vascularcomponent of the bleeding time. (34) These are described later in thesection entitled 'Newer Methods of Platelet Evaluation.'
Bone Marrow ExaminationExamination of the bone marrow can be performed to evaluate boththrombocytopenia or thrombocytosis, but it has little role in theevaluation of platelet dysfunction with a normal platelet count. Thebone marrow examination may be helpful to ascertain whetherthrombocytosis is due to reactive or myeloproliferative disorders.Thrombocytosis can often accompany iron deficiency; evaluation of serumiron studies together with the erythrocyte indices on the complete bloodcell count can usually diagnose the condition and obviate the need for abone marrow evaluation.
In a thrombocytopenic patient, when no otherreason for low platelet counts can be determined, the bone marrowexamination is useful for determining the presence or absence ofmegakaryocytes; absence indicates dysfunctional marrow, whereasincreased numbers suggest peripheral destruction with attempted bonemarrow compensation. Bone marrow examination can also detectmyelophthisic disorders, such as acute leukemia, lymphoma, or metastaticmalignancy, which could explain a patient's thrombocytopenia.Platelet AggregationPlatelet aggregation studies measure the ability of agonists tocause in vitro platelet activation and platelet-platelet binding.Platelet aggregation studies can be performed in whole blood by animpedance technique or in platelet-rich plasma by a turbidimetrictechnique. (35,36) Platelet aggregation techniques using a microtiterplate or flow cytometer have also been described, but are not widelyperformed. (37,38) Whole blood platelet aggregation can be combined withstudies of dense granule adenosine triphosphate release with alumiaggregometer.
(39) Turbidimetric platelet aggregation studiesrequire platelet-rich plasma prepared from a whole blood specimen. Manyfactors can affect the platelet aggregation results, such as theplatelet count, processing temperature, stirring rate, and processingtime (testing should be completed within 4 hours of phlebotomy). (40) Inaddition, clinicians who order the tests should advise patients todiscontinue using, if possible, any medication that may interfere withthe results of the test (Table 1).In the turbidimetric platelet aggregation assay, plateletaggregation is measured spectrophotometrically by the increase in lighttransmission after addition of an aggregation agonist. (35) The agoniststypically used in the assay include ADP, collagen, arachidonic acid, andepinephrine. Optimal platelet aggregation shows a biphasic pattern forthe agonists ADP and epinephrine; the initial increase in aggregation isdue to primary aggregation in response to activation of the glycoproteinIIb/IIIa platelet membrane receptor, whereas the second wave ofaggregation is the result of platelet degranulation with recruitment ofadditional platelet aggregates. Other agonists, such as arachidonicacid, thrombin receptor agonists, and collagen, usually show only asingle wave of aggregation.Another important reagent used in the evaluation of plateletfunction by aggregation is the antibiotic ristocetin, which facilitatesthe binding of VWF to the glycoprotein Ib/IX/V complex.
(41)Ristocetin-induced platelet aggregation evaluates aggregation after theaddition of various concentrations of ristocetin. This dose responseallows testing for both increased and decreased sensitivity toristocetin.
Most animals with hemostatic disorders present because of signs of excessive hemorrhage and diagnostic testing is most frequently performed in these patients. There are tests for each process of hemostasis (primary, secondary and fibrinolysis) as well as global tests of hemostasis that evaluate two or more of these hemostatic processes simultaneously, as well as the contribution of cells, such as viscoelastic methods or thromboelastography/thromboelastometry (TEG/TEM) and calibrated automated thrombography (CAT) or thrombin generation assays. Global tests require specific and dedicated equipment, which restricts their usage to academic institutions and large specialty practices. Hemostasis test algorithmThe results of screening tests dictate the need for further or more specific diagnostic testing or may prompt additional assays. The choice of assays (screening or otherwise) should be guided by knowledge of the patient (age, breed, sex, access to anticoagulant rodenticides, parasite exposure, type of bleeding symptoms, presence of underlying disease etc). If assay results are still not fruitful and the animal is still suspected of having a hemostatic disorder, referral of the patient to a hemostasis specialist would be worthwhile for more specialized or complex testing.
Referral of the actual patient versus referral of samples collected from the patient is recommended because some testing require the use of fresh platelets from the patient and cannot be done on stored or shipped samples, e.g. Platelet function assays. We have provided a suggested diagnostic testing algorithm for a bleeding patient as well as a table of expected test abnormalities with various disorders.
. Kandice Kottke-Marchant.
An Algorithmic Approach to Hemostasis Testing. CAP Press, Northfield, IL. ISBN: 978-0-930304-93-5. 380 Page Book that is ideal for Hematopathology Residents, but also certainly recommended for off-service residents with an interest in Coagulation. N.Key, M. O'Shaughnessy and D.Lillicrap. Practical Hemostasis and Thrombosis. Second Edition.
ISBN: 978-1-4051-8460-1. Somewhat smaller and less dense then the CAP book. 311 pages with excellent material but more focused on clinical hematology. May be more appropriate for Clinical Hematology fellows rotating through the lab.
S.T. Bennett, C.M. Laboratory Hemostasis: A Practical Guide for Pathologists. Springer, New York, NY. ISBN: 978-0387-36838-2. 238 page pocket book which is much less dense and detailed then the CAP book but focuses on laboratory issues in coagulation and is ideal for General Pathology residents doing their hematopathology rotation. C.
Practical Diagnosis of Hematologic Disorders. Volume 1. ASCP Press, Chicago, IL. ISBN: 0-89189-528-0. P.299-406. Coagulation section is succinct, and to the point. Not recommended as the only source for coagulation but not a bad starting point. A copy is available in the Bone Marrow Reading Room. If you wish to take it out please sign the register on the side of the west shelf.
